Key criteria for a CRO
Protocol flexibility is the first major consideration. How well does the system adapt to observational studies, randomized trials or complex protocols with multiple treatment arms? A rigid EDC forces compromises that can affect data quality.
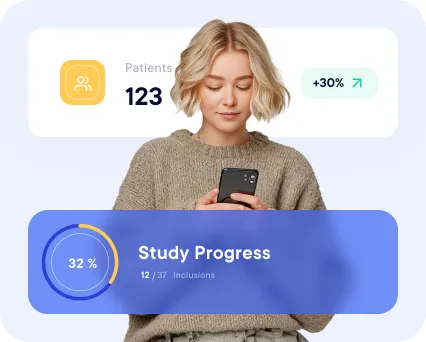
A multi-study monitoring dashboard allows project managers to visualize progress across their entire portfolio. This overview helps quickly identify delayed studies, underperforming sites or data quality issues that require intervention.
Ease of adoption has a direct impact on costs. Training a new project manager for three months on a complex system represents a significant investment. Intuitive interfaces reduce this burden and help teams become autonomous faster.
Best practices for implementation
Implementing an EDC system within a CRO requires a structured approach. Standardizing templates for similar studies accelerates future deployments. Creating reusable CRF libraries turns each new project into an assembly of proven components rather than a complete build from scratch.
Continuous team training ensures optimal use of eCRF features. An underused EDC system represents a wasted investment. High-performing CROs organize regular sessions to share best practices across projects.
Choosing the right solution for your organization
The best EDC for your CRO depends on your activity volume and positioning. Large international CROs tend to prioritize enterprise solutions such as Medidata or Veeva, accepting their complexity in exchange for advanced capabilities. Mid-sized CROs look for the right balance between robustness and agility, an area where Datacapt is effectively positioned. Smaller organizations often favor open-source solutions such as OpenClinica for their financial accessibility.
The final decision should consider not only current features, but also the system’s ability to evolve with your organization and with innovation in the sector.